Recently the Advisory Committee to the FDA recommended the approval of two new drugs (both are PCSK9 inhibitors that prevent the internalization of LDL receptors) to lower LDL cholesterol. Now the FDA has a chance to actually practice its real mandate, which is to protect the American public, not to generate new revenues for the drug companies.
Here are some very good reasons why a positive decision by the FDA to grant approval to these new drugs would represent a massive gamble for the American public.
What’s the goal?
The real target of any cardiovascular drug is to reduce heart disease. What’s the data on these drugs? There isn’t any (1,2). A reduction in a risk factor (i.e. LDL reduction) is not treating a disease. There are hundreds of known risk factors for heart disease (I personally like lowering inflammation as my favorite) besides elevated LDL cholesterol levels. At least the statins had some positive data in exceptionally high-risk male patients (but not female subjects) with existing heart disease that there was some benefit (3). However, in subsequent trials using overall mortality as an end point, the statins turn out to be rather pathetic drugs (4).
But statin defenders love to point out that they saved millions from death. Unfortunately, the data from the National Heart Lung and Blood Institute don’t back this up as shown in the following figure.
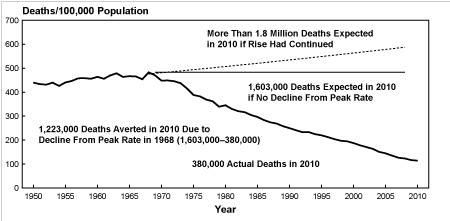
From 1950 to 1970 the death rate from heart disease was high. Then it started to drop in a straight-line fashion. It certainly wasn’t due to statins since they were not introduced until 1994. In fact, I am very hard pressed to see any change in the rate of decline. What could possibly explain such a dramatic reduction in mortality if it wasn’t the introduction of statins? How about starting with the Surgeon General’s Report in 1964 to stop smoking as shown in the graph below?
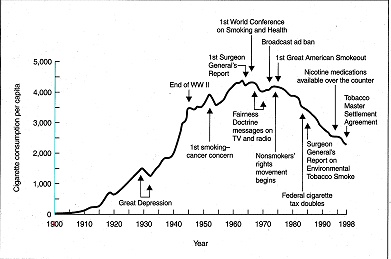
Heart disease was a rare disease at the turn of the century. It was only in 1920 that rates began to rise as shown in the figure below from the Centers of Disease Control. Unfortunately, the CDC didn’t separate classic coronary heart disease (CHD) from other diseases, such rheumatic fever, until 1952; but it appeared that CHD peaked in 1968, whereas stroke had been decreasing since 1900.
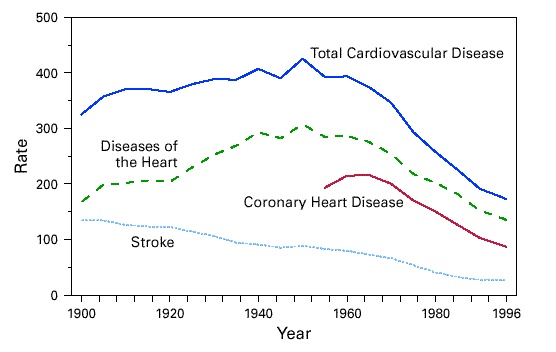
I don’t think you need a degree in statistics to see the rise and drop-off in heart disease mortality seems to be more highly correlated with changes in smoking than the introduction of statins.
Don’t Confuse Me with History
Using surrogate markers like lowering cholesterol instead using real clinical endpoints like death makes it easy for drug companies to manipulate the data. Statins were not the first drugs to lower cholesterol. Early drugs (like cholesyramine) also lowered cholesterol but increased all-cause mortality (5). Recent examples of other cholesterol-lowering drugs that didn’t decrease overall mortality include niacin (6), fibrates (7), CETP inhibitors (8), and cholesterol-absorption blocking drugs (9).
The reason statins have at least some benefits unlike other pure cholesterol-lowering drugs is because they have other effects (reducing inflammation is one) that also have benefits in treating heart disease. Nevertheless, statins remain totally ineffective in reducing all-cause mortality in women with or without pre-existing heart disease, very poor in preventing all-cause mortality in men without pre-existing heart disease (i.e. primary prevention), and not all that great in reducing all-cause mortality in men with existing heart disease (i.e. secondary prevention) (10,11). Considering the past track record with other cholesterol-lowering drugs and heart disease, I think it is only reasonable that the FDA should wait until a real clinical trial looking at real clinical outcomes on these new drugs is completed in 2017 to see if these drugs really work.
Do No Harm
These new drugs also have known neurocognitive problems (1,2). If people don’t have existing heart disease, then why treat them with new drugs that have other problems, especially cognitive ones? Likewise, no one seems to be concerned by the demonstrated ability of statins to increase diabetes by 9% (12). Since the best estimate is that you have to treat 200 people with statins for five years to prevent one heart attack, this also means that 18 of those 200 (9%) will likely develop diabetes. Not a very good trade-off.
Follow the Money Trail
There are hundreds of billions of easy sales with these new drugs. Who doesn’t want the newest of the new? But what if they don’t work according to the results of the 27,000-person trial to be completed in 2017? Will the drug companies return their profits to the insurance companies who paid for the drugs? Highly unlikely unless the FDA tells them to wait before selling an unproven drug to treat heart disease. References:
References:
- Sabatine MS, Giugliano RP, Wiviott SD, Raal FJ, Blom DJ, Robinson J, Ballantyne CM, Somaratne R, Legg J, Wasserman SM, Scott R, Koren MJ, and Stein EA. “Efficacy and safety of evolocumab in reducing lipids and cardiovascular events.” N Engl J Med 372(16):1500-1509 (2015).
- Robinson JG, Farnier M, Krempf M, Bergeron J, Luc G, Averna M, Stroes ES, Langslet G, Raal FJ, El Shahawy M, Koren MJ, Lepor NE, Lorenzato C, Pordy R, Chaudhari U, and Kastelein JJ. “Efficacy and safety of alirocumab in reducing lipids and cardiovascular events.” N Engl J Med 372:1489-1499 (2015).
- 4S Investigators. “Randomised trial of cholesterol lowering in 4444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S).” Lancet 344:1383-1389 (1994).
- Ray KK, Seshasai SR, Erqou S, Sever P, Jukema JW, Ford I, and Sattar N. “Statins and all-cause mortality in high-risk primary prevention: a meta-analysis of 11 randomized controlled trials involving 65,229 participants.” Arch Intern Med 170:1024-1031 (2010).
- The Lipid Research Clinics Investigators. “The Lipid Research Clinics Coronary Primary Prevention Trial. Results of 6 years of post-trial follow-up.” Arch Intern Med 152:1399-1410 (1992).
- Boden WE, Probstfield JL, Anderson T, Chaitman BR, Desvignes-Nickens P, Koprowicz K, McBride R, Teo K, and Weintraub W. “Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. “ N Engl J Med 365:2255-22567 (2011).
- Ginsberg HN, Elam MB, Lovato LC, Crouse JR, Leiter LA, Linz P, Friedewald WT, Buse JB, Gerstein HC, Probstfield J, Grimm RH, Ismail-Beigi F, Bigger JT, Goff DC, Cushman WC, Simons-Morton DG, and Byington RP. “Effects of combination lipid therapy in type 2 diabetes mellitus.” N Engl J Med 362:1563-1574 (2010).
- Nissen SE, Tardif JC, Nicholls SJ, Revkin JH, Shear CL, Duggan WT, Ruzyllo W, Bachinsky WB, Lasala GP, Lasala GP, and Tuzcu EM. “Effect of torcetrapib on the progression of coronary atherosclerosis.” N Engl J Med 356:1304-1316 (2007).
- Prasad V and Vandross A. “Cardiovascular primary prevention.” Arch Intern Med 172:656-659 (2012).
- Kendrick M. The Great Cholesterol Con. John Blake. London (2007).
- Prasad V. “Statins, primary prevention and overall mortality.” Ann Intern Med 160:867-869 (2014).
- Sattar N, Preiss D, Murray HM, Welsh P, Buckley BM, de Craen AJ, Seshasai SR, McMurray JJ, Freeman DJ, Jukema JW, Macfarlane PW, Packard CJ, Stott DJ,Westendorp RG, Shepherd J, Davis BR, Pressel SL, Marchioli R, Marfisi RM, Maggioni AP, Tavazzi L, Tognoni G, Kjekshus J, Pedersen TR, Cook TJ, Gotto AM, Clearfield MB, Downs JR, Nakamura H, Ohashi Y, Mizuno K, Ray KK, and Ford I. “Statins and risk of incident diabetes: a collaborative meta-analysis of randomised statin trials.” Lancet 375:735742 (2010).






Let Us Know What You Thought about this Post.
Put your Comment Below.